 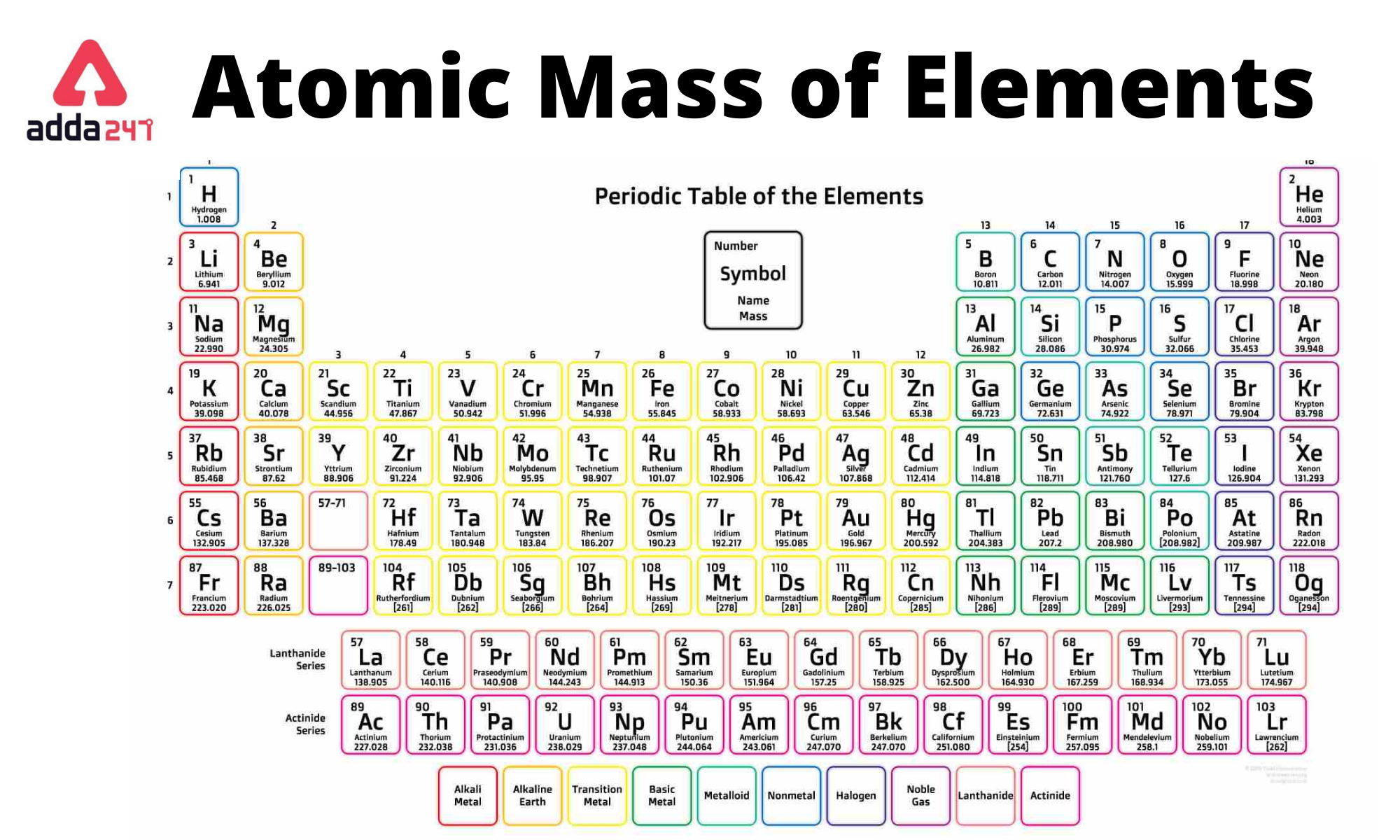
Metallic character decreases from left to right in each period of the periodic table. Metallic character increases down a group. Going down a group, the atomic radius increases: the outer electrons are farther from the nucleus and are easier to remove, thus increasing the metallic character.The metallic character is essentially the ease with which an atom gives up an electron Metalloid: an element with properties intermediate between metals and nonmetals. Metalloids divide the periodic table in a zigzag line between metals on the left and nonmetals on the right.Nonmetal: poor conductor of heat and electricity. They are generally softer elements, often colored, with lower melting and boiling points than most metals.Metal: good conductor of heat and electricity. They tend to be hard, metallic-looking solids with high melting and boiling points. The majority of elements are metals.Most elements can be classified as metals or nonmetals based on their ability to conduct heat and electricity: ⇒ very stable elements, generally considered to be inert gases Noble gases: last column = helium, neon, argon, (krypton, xenon, radon).⇒ colorful and corrosive nonmetals, very reactive  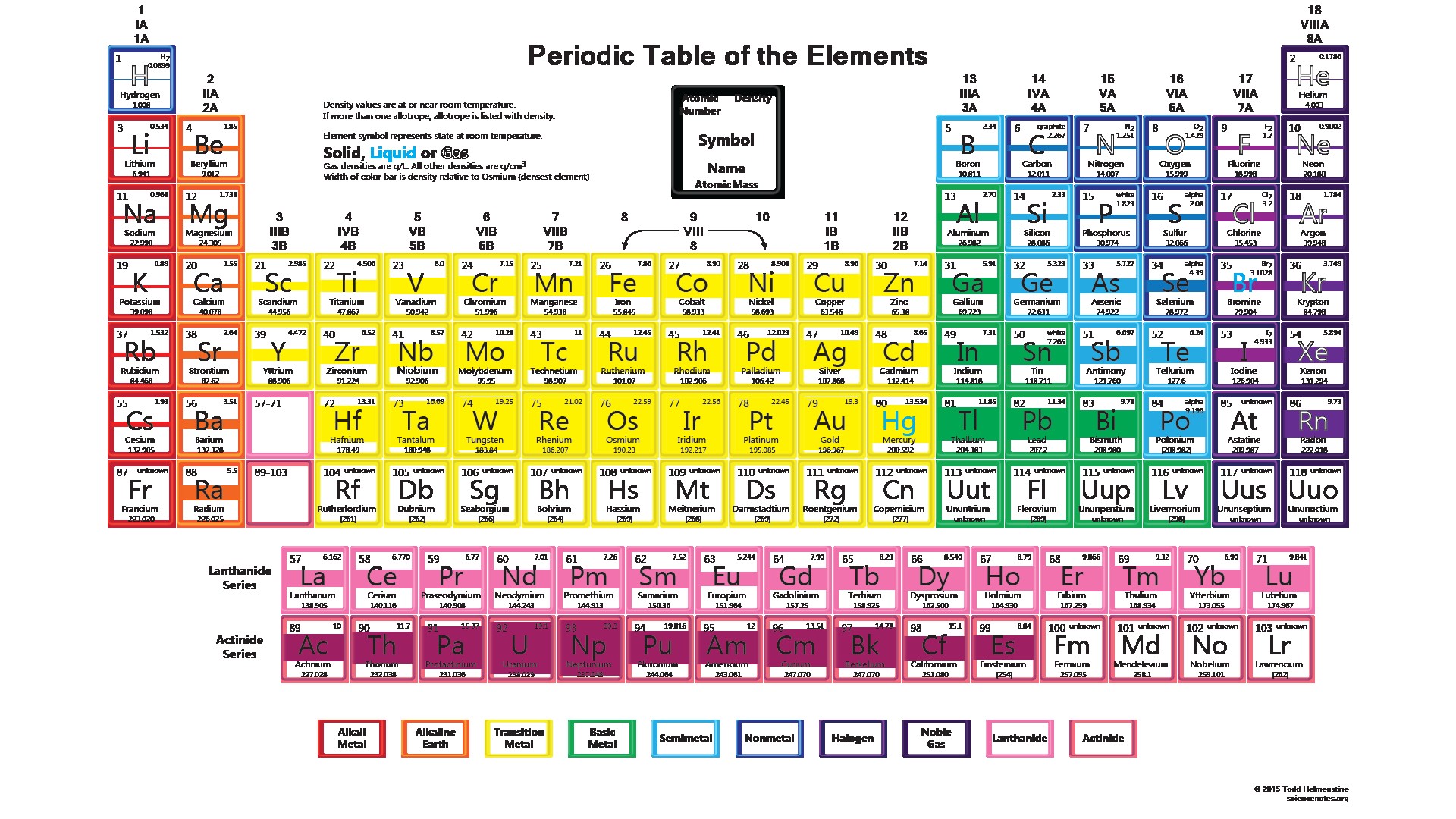
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
/PeriodicTableSigFig-NoBG-56a12da75f9b58b7d0bcd00f.png)
 RSS Feed
RSS Feed
